Tritium Radiation: Essential Facts You Need to Know Today
from web site
Have you ever wondered what powers the glowing lights in emergency exit signs or watch dials that shine in the dark? The answer lies in something called Tritium radiation.
Understanding Tritium radiation isn’t just for scientists—it’s important for you too. Whether you’re curious about safety, technology, or how certain everyday items work, knowing the basics of Tritium radiation can help you make smarter choices. Keep reading, and you’ll discover what Tritium radiation is, how it affects you, and why it matters more than you might think.

Basics Of Tritium
Tritium is a rare form of hydrogen. It holds unique traits that make it important in science and industry. Understanding the basics of tritium helps us see its role in energy, research, and safety.
This section breaks down tritium into simple parts. It explains what tritium is, where it comes from, and its key physical features. Each part gives a clear view of this special element.
What Is Tritium?
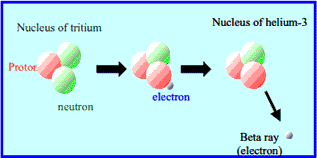
Tritium is a radioactive isotope of hydrogen. Its nucleus has one proton and two neutrons. This makes it heavier than normal hydrogen. Tritium emits weak radiation and decays over time. It is used in glow-in-the-dark devices and nuclear fusion research.
Natural Vs Artificial Sources
Tritium occurs naturally in the upper atmosphere. Cosmic rays hit nitrogen atoms and create tritium. This natural tritium falls to Earth with rainwater. Artificial tritium is made in nuclear reactors. It is produced for scientific and military uses. Natural tritium levels are very low compared to man-made sources.
Physical Properties
Tritium is a colorless, odorless gas at room temperature. It behaves like normal hydrogen gas but is heavier. Its radioactivity makes it glow faintly in the dark. Tritium’s half-life is about 12 years. This means it loses half its radioactivity in that time. It combines easily with oxygen to form tritiated water.
Radiation Characteristics
Tritium radiation has unique traits that define its behavior and impact. Understanding these traits helps explain its uses and safety. Its radiation comes from a natural process inside the atom. The way it decays and the strength of its radiation are important to know. These features make tritium useful in many fields.
Type Of Radiation Emitted
Tritium emits beta radiation. Beta particles are tiny and fast electrons. They come from the nucleus of the tritium atom. This radiation is low energy and weak. It cannot pass through thick materials. Even a sheet of paper or skin stops it easily. This makes tritium safer than other radioactive materials.
Half-life And Decay Process
Tritium has a half-life of about 12.3 years. Half-life means the time for half of the atoms to decay. During tritium technologies , tritium changes into helium-3. This change releases beta particles. The process is slow and steady. It means tritium keeps emitting radiation for many years. This steady decay suits long-term uses.
Radiation Intensity And Penetration
Tritium’s radiation intensity is very low. The beta particles it emits cannot travel far. They only move a few millimeters in air. They do not penetrate the skin deeply. Tritium poses little risk outside the body. Inside the body, it can be harmful if inhaled or swallowed. This low penetration makes tritium useful for glow-in-the-dark items.
Uses Of Tritium
Tritium is a special type of hydrogen with a small amount of radioactivity. It has many important uses across different fields. People use tritium in ways that help science, industry, and safety. Its unique properties make it valuable for many tasks.
Below are some common uses of tritium in medicine, industry, and the military.
Medical Applications
Tritium helps doctors study how drugs work in the body. It acts as a marker in medical tests. Scientists use tritium to track cells and chemicals in research. This helps find better treatments for diseases. Tritium also supports imaging techniques that show details inside the body.
Industrial Uses
Factories use tritium to make glowing signs and watches. Its light does not need electricity or batteries. Tritium is also in safety devices like exit signs and emergency lights. It helps workers see clearly in dark or smoky places. Tritium’s steady glow improves safety in many settings.
Military And Aerospace
The military uses tritium for night-vision instruments and weapon sights. It helps soldiers see targets in the dark. In aerospace, tritium powers light sources for aircraft controls. Its glow works well in low-light conditions. Tritium supports missions by improving visibility and safety.

Health And Safety Concerns
Tritium is a radioactive form of hydrogen used in various industries. Its health and safety concerns matter a lot. Understanding these concerns helps protect people and the environment. This section covers key points about exposure risks, safety rules, and how to handle tritium safely.
Exposure Risks
Tritium emits low-energy beta radiation. It cannot penetrate the skin. The main risk is breathing or swallowing tritium. Once inside the body, it can affect cells. High amounts may cause harm over time. People near tritium sources should avoid direct contact. Proper protection reduces exposure risks.
Safety Standards And Regulations
Governments set strict rules for tritium use. These rules limit how much tritium workers can handle. Regulatory agencies monitor tritium levels in workplaces. They require regular safety checks and training. Following these standards keeps workers safe. Companies must report tritium use and any leaks.
Handling And Storage Precautions
Tritium must be stored in sealed containers. Containers prevent tritium from escaping into the air. Handling tritium needs special tools and gear. Workers wear gloves and masks to avoid inhaling it. Work areas should have good ventilation. Safe storage and handling prevent accidents and exposure.
Environmental Impact
Tritium radiation affects the environment in several ways. It is important to understand how it moves and changes in nature. This helps us manage risks and protect ecosystems. The main concerns involve water, air, living organisms, and waste handling.
Tritium In Water And Air
Tritium can enter water sources through leaks or discharges. It mixes easily with water because it forms tritiated water. This water moves through rivers, lakes, and groundwater. Tritium in the air comes from evaporation or gas release. It can spread over large areas and enter the atmosphere. Monitoring tritium levels in water and air is crucial for safety.
Bioaccumulation Potential
Tritium does not build up much in plants or animals. It usually leaves the body quickly through natural processes. This means it has a low bioaccumulation risk. Yet, small amounts can still enter food chains. Scientists study these effects to understand any long-term impact on wildlife and humans.
Waste Management
Proper waste management reduces tritium release into the environment. Facilities store and treat tritium-containing waste carefully. They use barriers and controlled disposal methods. This limits contamination of soil, water, and air. Good waste practices help protect communities and nature from harm.
Detection And Measurement
Detecting and measuring tritium radiation is essential for safety and environmental monitoring. Tritium is a weak radioactive substance and requires special tools to detect it. The process helps identify radiation levels and prevent exposure risks.
Understanding how to detect and measure tritium radiation allows for better control and protection. The methods used are designed to capture low-energy beta particles emitted by tritium. Accurate measurement ensures the environment and people remain safe.
Common Detection Methods
Tritium detection often uses liquid scintillation counting. This method captures beta particles in a liquid solution. Another method is using gas proportional counters that detect radiation in the air. Sometimes, solid-state detectors are used for surface contamination checks. Each method suits different environments and detection needs.
Measuring Radiation Levels
Radiation levels from tritium are measured in becquerels or curies. These units show the number of disintegrations per second. Instruments record counts that technicians convert into radiation levels. Proper calibration of devices is crucial for accurate results. Measurements must be taken carefully to avoid errors.
Interpreting Results
Understanding the results helps decide on safety actions. Low levels of tritium usually mean no immediate danger. Higher levels may require cleanup or restricted access. Interpreting data also involves comparing measurements to legal limits. Clear interpretation keeps people and the environment protected.
Future Trends
The future of tritium radiation holds promising changes. These changes focus on safer use, new technology, and clearer rules. Progress in these areas can benefit many fields. Industries could use tritium more wisely and with less risk. Understanding what comes next is key for users and regulators.
Advances In Tritium Use
Scientists explore new ways to use tritium. One area is in medical treatments that need precise radiation. Another is in improving energy sources with better efficiency. Research aims to reduce waste and increase tritium's life span. These advances could make tritium more practical and eco-friendly.
Improved Safety Technologies
Safety remains a top priority in tritium use. New devices can detect leaks faster and with more accuracy. Protective materials are evolving to better contain radiation. These tools help prevent accidents and reduce exposure risks. Safer technology builds trust among workers and the public.
Regulatory Developments
Rules governing tritium are becoming clearer and stricter. Governments work to balance innovation with safety. Updated regulations guide proper handling and disposal. They also set limits to protect health and environment. Strong regulations support responsible use of tritium worldwide.
Frequently Asked Questions
What Is Tritium Radiation And Its Main Uses?
Tritium radiation comes from a radioactive hydrogen isotope. It's used in self-luminous devices, watches, and nuclear fusion research due to its low energy and safe handling.
Is Tritium Radiation Harmful To Humans?
Tritium emits weak beta radiation, which can't penetrate skin. However, ingestion or inhalation can pose health risks, so proper safety measures are essential.
How Is Tritium Radiation Detected And Measured?
Tritium radiation is detected using liquid scintillation counters or gas proportional counters. These devices measure the beta particles emitted by tritium decay accurately.
Where Is Tritium Naturally Found In The Environment?
Tritium occurs naturally in trace amounts in the atmosphere and water. It forms when cosmic rays interact with atmospheric gases, maintaining low environmental levels.
Conclusion
Tritium radiation plays a role in many industries today. It emits low energy, making it safer than other radiation types. People use tritium in watches, signs, and scientific tools. Understanding its properties helps us handle it carefully. Safe use and proper disposal protect the environment and health.
Learning about tritium radiation shows how science affects daily life. Keep exploring to know more about this unique element.
