The Ultimate Guide To CCPL COVID-19 Test Kit Distribution - Chesterfield County
from web site
How Coronavirus (COVID-19) Testing - HHS.gov can Save You Time, Stress, and Money.
More information on this federal program is expected quickly. For people covered by initial fee-for-service Medicare, Medicare pays for COVID-19 diagnostic tests carried out by a laboratory, such as PCR and antigen tests, without any beneficiary expense sharing when the test is ordered by a physician, non-physician professional, pharmacist, or other authorized healthcare professional.
At this time initial Medicare can not pay for at-home tests through this program. Medicare Advantage plans may provide protection and payment for at-home over the counter COVID-19 tests, so customers covered by Medicare Benefit should contact their plan. In accordance with the American Rescue Strategy, State Medicaid and CHIP programs are needed to cover FDA-authorized at-home COVID-19 tests.
We appreciate your understanding as our associates work all the time to support you.

Engineering Health and medication Press release Research Science January 24, 2022 The Consistency COVID-19 test includes a little, low-cost detector (left) that can processes four reaction tubes shown in the detector and a sample collection device (center). Outcomes are shown on a smart device (right). Mark Stone/University of Washington Researchers at the University of Washington have established a new test for COVID-19 that combines the speed of over the counter antigen tests with the precision of PCR tests that are processed in medical labs and hospitals.
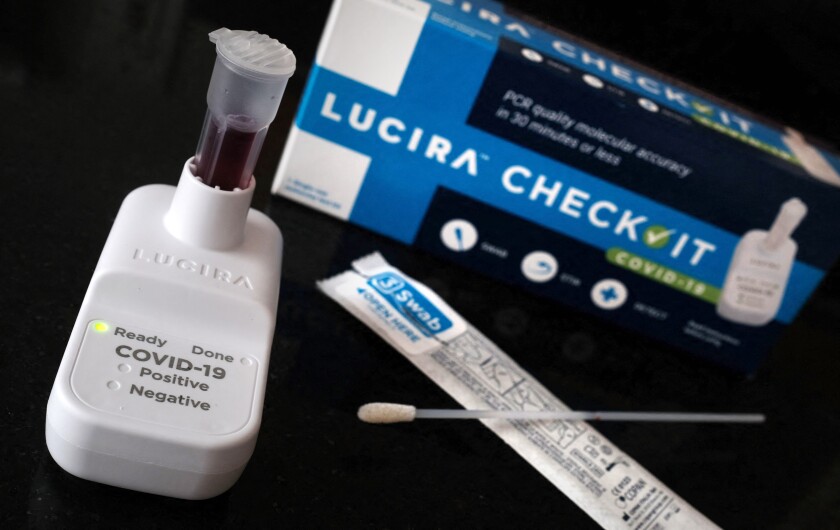
The Definitive Guide to iHealth COVID-19 Antigen Rapid Test (Single Box with 2 Tests)
However whereas traditional PCR tests can take a number of hours, the Harmony package can offer outcomes in less than 20 minutes for some samples and with similar accuracy. "We designed the test to be low-cost and easy enough that it might be used anywhere," said Barry Lutz, a UW associate teacher of bioengineering and investigator with the Brotman Baty Institute for Accuracy Medication.
15 in Science Advances that describes the Consistency COVID-19 test package. The researchers established Consistency to be basic and user friendly, using ready-to-use reagents. The test uses a "PCR-like" method to spot the presence of the SARS-Co, V-2 RNA genome in a nasal swab sample with the aid of a little, affordable detector, which was likewise developed by Lutz's group.
The detector can handle as much as four samples at a time and would fit into a standard cars and truck's glove compartment. In Check it Out , lead author Nuttada Panpradist, then a UW doctoral student in bioengineering, operates in the laboratory on a model of the Consistency COVID-19 test. Mark Stone/University of Washington The precision of COVID-19 tests has actually been a pushing matter throughout the pandemic.
